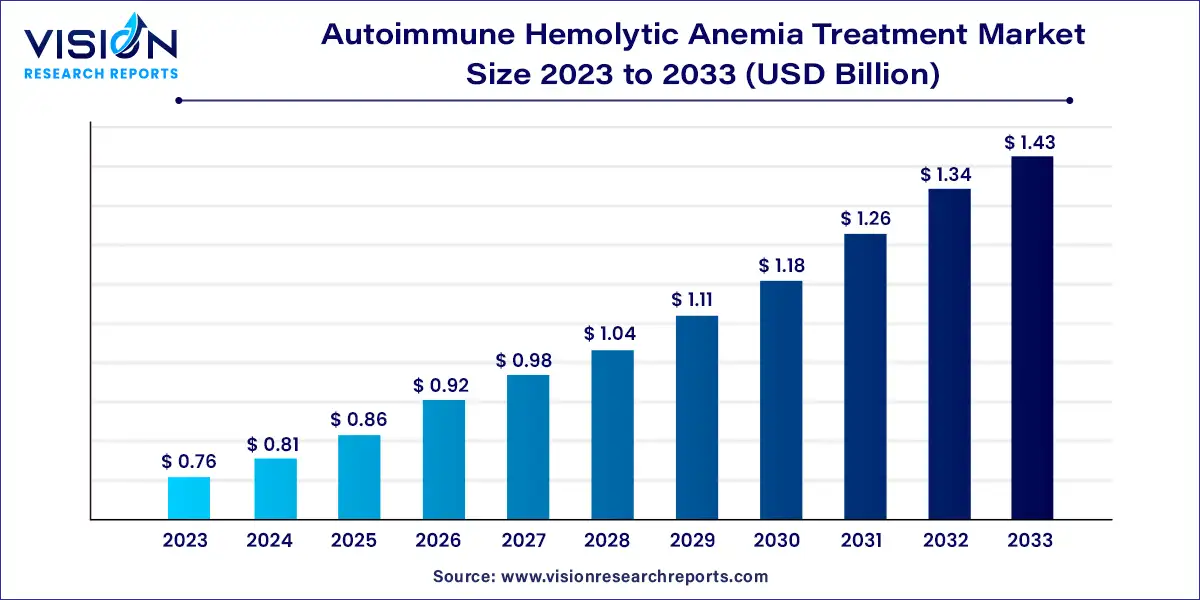
The global autoimmune hemolytic anemia treatment market size was estimated at around USD 0.76 billion in 2023 and it is projected to hit around USD 1.43 billion by 2033, growing at a CAGR of 6.53% from 2024 to 2033.
Autoimmune Hemolytic Anemia (AIHA) is a rare but serious condition where the immune system mistakenly attacks and destroys red blood cells, leading to hemolysis and subsequent anemia. This condition necessitates prompt and effective treatment to manage symptoms and prevent complications. The AIHA treatment market has been evolving, driven by advances in medical research, increased awareness, and the development of innovative therapeutic approaches.
The growth of the autoimmune hemolytic anemia (AIHA) treatment market is driven by an advancements in medical research have led to the development of novel therapies, such as biologics and targeted treatments, which offer more effective options for patients who do not respond to traditional methods. Secondly, increasing awareness and improved diagnostic techniques are leading to earlier and more accurate detection of AIHA, thus expanding the patient pool requiring treatment. Additionally, regulatory incentives for orphan drug development are encouraging pharmaceutical companies to invest in AIHA research. Lastly, the expanding healthcare infrastructure in emerging markets is improving access to advanced treatments, further fueling market growth.
The U.S. autoimmune hemolytic anemia treatment market size was estimated at USD 0.19 billion in 2023 and it is expected to surpass around USD 0.37 billion by 2033, poised to grow at a CAGR of 6.53% from 2024 to 2033.
The U.S. market is projected to grow at a lucrative rate due to the high incidence of AIHA, better reimbursement policies, an improved healthcare infrastructure, and favorable initiatives by government and non-government bodies aimed at enhancing healthcare services for AIHA patients. According to the American Society of Hematology, the prevalence of AIHA per million is approximately 190 in Medicare claims, 70 in Optum claims, and 50 in other claims.
In 2023, North America accounted for the largest market share of 37% and is expected to maintain its dominance throughout the forecast period. Factors contributing to this growth include the high incidence of autoimmune hemolytic anemia, the presence of leading pharmaceutical companies involved in developing and marketing novel therapeutic drugs, and favorable government initiatives. For instance, the U.S. FDA approved Enjaymo (sutimlimab) for treating hemolysis in adults with cold agglutinin disease (CAD) in February 2022. Such initiatives are expected to sustain the region's leading position.
The Asia Pacific market is expected to witness the fastest growth over the forecast period. The region's large target population, high unmet medical needs, and a burgeoning healthcare industry present significant growth potential. Countries like Japan, China, and India, with their large patient bases and unmet medical needs, offer substantial market opportunities for key players. Additionally, increasing R&D activities in the region are expected to drive market growth. For example, in October 2022, HUTCHMED Limited initiated a phase 2/3 trial of sovleplenib in adult patients with warm antibody autoimmune hemolytic anemia in China.
Warm autoimmune hemolytic anemia segment led the market with a share of 69% in 2023 and is expected to maintain its dominance over the forecast period. High share of the segment can be attributed to the growing prevalence of the disease, increasing treatment rate, and growing awareness among people. For instance, warm autoimmune hemolytic anemia (wAIHA) accounts for around 60% to 70% of total autoimmune hemolytic anemia cases. Moreover, the incidence of wAIHA is around 1 in 35,000 and 1 in 80,000 people in North America and Western Europe, respectively.
Cold autoimmune hemolytic anemia segment is anticipated to grow at the fastest rate over the forecast period owing to the increasing incidence of disease, strong research & development activities, and increasing approval of novel drugs for the disease indication. For instance, Sanofi developed Enjaymo (sutimlimab) for the treatment of cold agglutinin disease and has received market approval from the U.S. FDA, European Commission, and Japanese Ministry of Health, Labor and Welfare. Moreover, people with cold AIHA may experience fatigue, acute hemolytic crisis, and other complications.
Corticosteroids led the autoimmune hemolytic anemia treatment market with a 59% share in 2023, supported by high prescription rates of drugs like hydrocortisone and prednisone and their effective treatment outcomes. Corticosteroids are the first-line therapy for AIHA, with over 65% of patients receiving them to suppress the immune response. Immunosuppressive agents are the second most common drug class used.
However, the segment for other drug classes is expected to grow the fastest. This growth is driven by ongoing R&D into novel drug classes, such as biological drugs, which offer improved results, and the introduction of new drugs by key market players. In 2022, the U.S. FDA, European Commission, and Japan's regulatory authorities approved sutimlimab for treating cold AIHA. The anticipated commercial launches of investigational biological drugs are expected to further boost this segment's growth.
Injectable drugs dominated the market with a 64% share in 2023 and are projected to remain the leading segment. The high market penetration of injectable drugs, including corticosteroids, folic acids, and emerging biological drugs, along with several pharmaceutical companies offering corticosteroids, supports this segment. Increased R&D activities for novel biological therapeutics and approvals of injectable drugs are expected to drive growth.
Oral drugs are anticipated to show the fastest growth rate during the forecast period. Factors such as the increasing adoption of immunosuppressive agents, patient convenience, and patient-centric approaches are driving this growth. Immunosuppressive agents, accounting for around 30% of total prescriptions in AIHA treatment, are the second most common drug class used
Hospital pharmacies led the market with a 61% share in 2023, driven by the high hospitalization rate due to hemolytic anemia, especially among the elderly. The severity and high mortality rate of AIHA have increased hospitalization rates, significantly boosting this segment's share.
The online pharmacy segment is anticipated to expand at the fastest rate over the forecast period. This growth is driven by the increasing use of the internet and smartphones, the convenience of ordering medications through e-commerce platforms, and the rise in global e-commerce services offering medicinal products. Additionally, the growing adoption of telemedicine for consultations, diagnostic services, and obtaining medications is expected to further bolster this segment's growth in the coming years.
By Type
By Drug Class
By Route of Administration
By Distribution Channel
By Region
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others