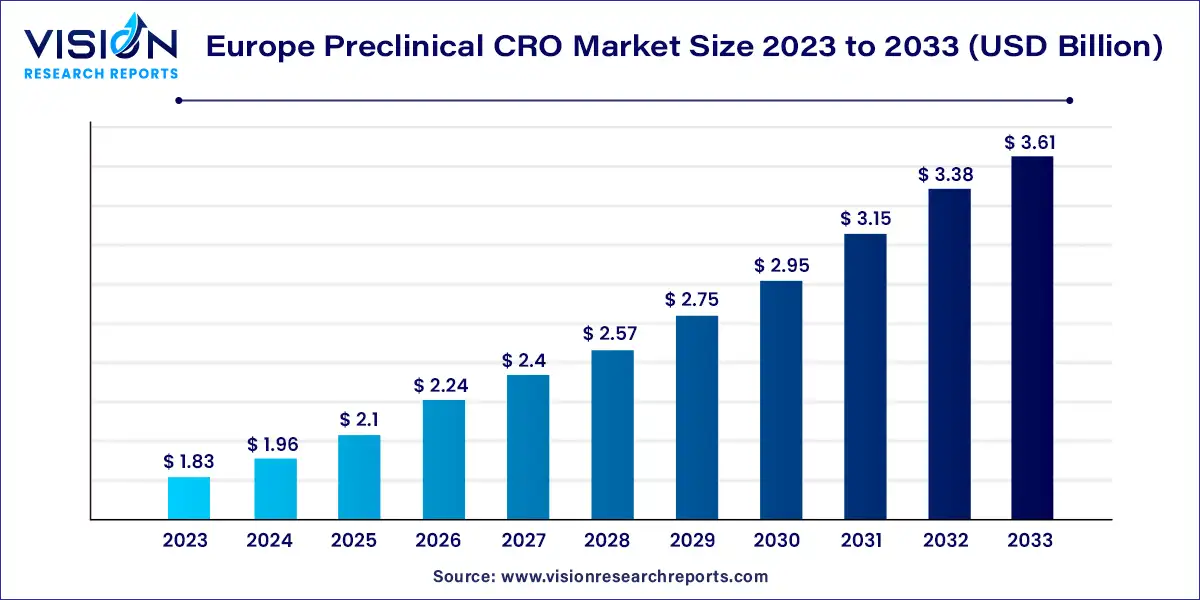
The Europe preclinical CRO market size was estimated at USD 1.83 billion in 2023 and it is expected to surpass around USD 3.61 billion by 2033, poised to grow at a CAGR of 7.04% from 2024 to 2033.
The Europe preclinical contract research organization (CRO) market is a dynamic sector integral to pharmaceutical and biotechnology advancements. As a pivotal component of drug development, preclinical CROs provide essential services ranging from pharmacology and toxicology studies to efficacy assessments.
The Europe preclinical Contract Research Organization (CRO) market is experiencing significant growth driven by an increasing outsourcing by pharmaceutical companies to streamline drug development processes and access specialized expertise is a major driver. Additionally, the rise in biopharmaceuticals necessitates stringent preclinical testing, further boosting demand for CRO services. Regulatory pressures also play a crucial role, pushing companies towards outsourcing to ensure compliance and expedite time-to-market for new therapies. These factors collectively contribute to the expanding landscape of the Europe preclinical CRO market, promising continued growth and innovation in pharmaceutical research and development.
In 2023, Patient-Derived Organoid (PDO) models emerged as market leaders, capturing a substantial 81% revenue share. These models are pivotal for studying personalized patient responses to therapies, facilitating advancements in precision medicine. PDOs are noted for their ease of cultivation and versatility in researching various diseases, particularly cancer, thereby driving growth in this segment.
Patient-Derived Xenograft (PDX) models are anticipated to witness robust growth in the coming years. This growth is fueled by increasing demand for personalized medicine, advancements in PDX technology, and heightened investments in cancer research. Moreover, the need for more accurate and predictive cancer models for drug development, along with the emphasis on humanized models, further contributes to this trend.
In 2023, the biopharmaceutical companies segment held the largest revenue share at 81%. This growth is attributed to the trend of outsourcing services, rapid technological advancements, and urbanization trends. The demand for cost-effective and time-efficient drug development processes also plays a significant role, as biotechnology and biopharmaceutical firms intensify their R&D efforts.
The government and academic institutes segment also commanded a notable revenue share in 2023, driven by increasing outsourcing of preclinical services to Contract Research Organizations (CROs). Academic institutions and government bodies play critical roles in early-stage research and development, relying on CROs for specialized expertise and efficient services, thereby stimulating market expansion.
In 2023, the toxicology testing segment led the market with a substantial 27% revenue share. Stringent regulatory requirements for safety and efficacy assessments during drug development bolstered the segment's growth. Additionally, the presence of established biopharmaceutical companies and specialized CROs focusing on early drug discovery further supported market expansion.
Bioanalysis and DMPK (Drug Metabolism and Pharmacokinetics) studies also held significant market shares in 2023. The increasing demand for pharmacokinetic services, particularly in supporting toxicology studies for IND-enabling evaluations, drove growth in this segment. The growing importance of DMPK in assessing drug properties across the drug development lifecycle contributed to its expanding role in the market.
In 2023, the UK dominated the European preclinical CRO market with a leading market share of 26%. This growth is attributed to substantial R&D investments, the presence of major pharmaceutical giants like GlaxoSmithKline plc and AstraZeneca, and the local presence of prominent early-stage development CROs such as Charles River Laboratories and Envigo. The UK's robust regulatory framework overseen by the Medicines and Healthcare Products Regulatory Agency (MHRA) ensures stringent guidelines for preclinical trials, safeguarding data integrity and quality.
Germany's preclinical CRO market experienced significant growth in 2023, driven by technological advancements, high-quality clinical resources, government initiatives supporting clinical research, and substantial R&D investments. The country's pharmaceutical sector, renowned for its research capabilities, coupled with numerous clinical trials supported by research-oriented pharmaceutical companies, further bolstered market expansion.
In March 2024, ALS Limited acquired the remaining 51% stake in Nuvisan, a European CRO, at no cost. Nuvisan, which generated USD 245 million in revenue in 2023, specializes in preclinical and clinical development, as well as drug discovery services. The acquisition aims to boost earnings growth and shareholder value, leveraging Nuvisan's strategic footprint in Western Europe and its expertise in pharmaceutical research and development.
By Service
By Model Type
By End-use
By Country
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others