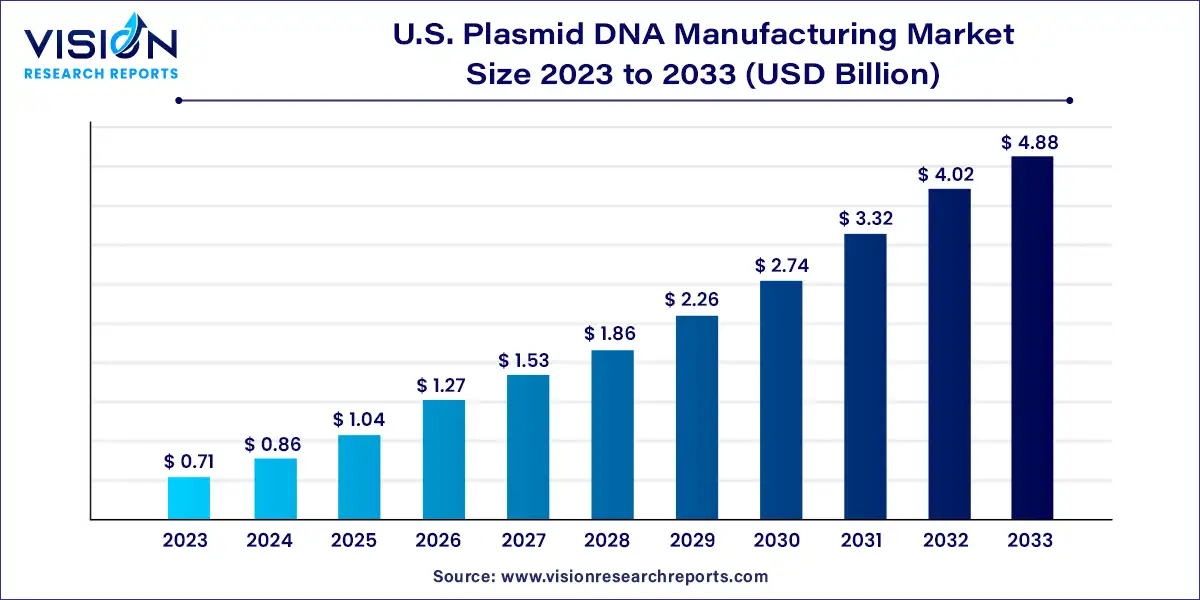
The U.S. plasmid DNA manufacturing market size was surpassed at USD 0.71 billion in 2023 and is expected to hit around USD 4.88 billion by 2033, growing at a CAGR of 21.25% from 2024 to 2033.
The U.S. Plasmid DNA manufacturing market stands at the forefront of biotechnological advancements, playing a pivotal role in various fields such as gene therapy, vaccine development, and molecular biology research.
The growth of the U.S. Plasmid DNA manufacturing market is propelled by an increasing prevalence of genetic disorders has spurred demand for gene therapy and personalized medicine solutions, driving the need for plasmid DNA manufacturing. Additionally, advancements in gene editing technologies, such as CRISPR-Cas9, have expanded the scope of applications for plasmid DNA, particularly in the development of novel therapeutics and vaccines. Furthermore, robust investments in research and development by biopharmaceutical companies and academic institutions have accelerated innovation in plasmid DNA manufacturing processes, leading to improved scalability, efficiency, and cost-effectiveness.
In 2023, the cell and gene therapy sector dominated the market, holding a substantial 56% share. This was primarily attributed to the widespread adoption of gene therapy in treating various genetically inherited diseases. The increasing utilization of cell and gene therapy-based medications is fueled by advancements in next-generation transfer vectors and plasmid DNA technology.
Furthermore, the DNA vaccines segment is poised to experience the highest compound annual growth rate (CAGR) from 2024 to 2033. The growing demand for DNA vaccines is attributed to their simple manufacturing process, which can be seamlessly integrated into the production of traditional vaccines in large-scale manufacturing facilities. Additionally, numerous plasmid DNA and viral vectors are undergoing exploration for human applications due to their inherent advantages. These investigations aim to evaluate their potential and expedite the development of vaccines.
In 2023, the cancer segment dominated the market with a substantial 42% share. This was attributed to the increasing incidence of cancer cases in the U.S., largely due to poor dietary habits, high alcohol consumption, smoking, and lack of physical activity. Despite the effectiveness of various cancer therapies, cancer-related fatalities remain prevalent, constituting a significant portion of mortality cases.
Furthermore, the genetic disorder segment is projected to witness significant growth from 2024 to 2033. Numerous private and public companies in the U.S. are focusing on this disease segment to develop advanced gene therapy treatments. With over 1500 ongoing clinical trials in the U.S. targeting genetic disorders, gene therapy in this domain emerges as a pivotal segment in the market during the forecast period.
In 2023, the clinical therapeutics segment emerged as the market leader, capturing a substantial 56% share. Plasmid DNA has gained prominence in clinical research endeavors, particularly with the advent of genetic vaccination techniques and personalized treatment approaches. Moreover, numerous therapies targeting life-threatening ailments such as HIV, HPV, and cancer are undergoing clinical trials, underscoring the pivotal role of plasmid DNA. For instance, in October 2022, the Ludwig Institute for Cancer Research concluded a phase I clinical trial of NY-ESO-1 Plasmid DNA for treating bladder, prostate, esophageal, non-small cell lung cancers, and sarcomas.
Meanwhile, the pre-clinical segment is poised to exhibit the highest compound annual growth rate (CAGR) from 2024 to 2033. Plasmid DNA finds extensive utility in pre-clinical trials during the research and development (R&D) phase, particularly in toxicology and bio-distribution studies. Consequently, the market for pre-clinical plasmid DNA therapeutics is expected to witness notable expansion over the forecast period, driven by a robust development pipeline for gene therapy and vaccines.
In 2023, the GMP grade segment emerged as the dominant force, capturing a significant 86% market share. This surge was driven by the escalating clinical developments in cell and gene therapy, along with DNA vaccines, coupled with the continuous approvals of newly marketed therapies. Additionally, stringent regulations governing the Good Manufacturing Practice (GMP) manufacturing of plasmids have provided further impetus to advanced therapy developers.
Meanwhile, the R&D grade segment is poised to experience the highest compound annual growth rate (CAGR) from 2024 to 2033. Plasmid DNA has garnered considerable attention due to its increasing demand and wide-ranging applications. The extensive use of R&D grade plasmid DNA in research settings, as well as in the fields of drug development and discovery by therapy developers, contributes significantly to its market demand. Moreover, advancements in manufacturing facilities and technology are anticipated to drive the adoption rate of R&D-grade plasmid DNAs throughout the forecast period.
By Grade
By Development Phase
By Application
By Disease
 Cross-segment Market Size and Analysis for
Mentioned Segments
Cross-segment Market Size and Analysis for
Mentioned Segments
 Additional Company Profiles (Upto 5 With No Cost)
Additional Company Profiles (Upto 5 With No Cost)
 Additional Countries (Apart From Mentioned Countries)
Additional Countries (Apart From Mentioned Countries)
 Country/Region-specific Report
Country/Region-specific Report
 Go To Market Strategy
Go To Market Strategy
 Region Specific Market Dynamics
Region Specific Market Dynamics Region Level Market Share
Region Level Market Share Import Export Analysis
Import Export Analysis Production Analysis
Production Analysis Others
Others